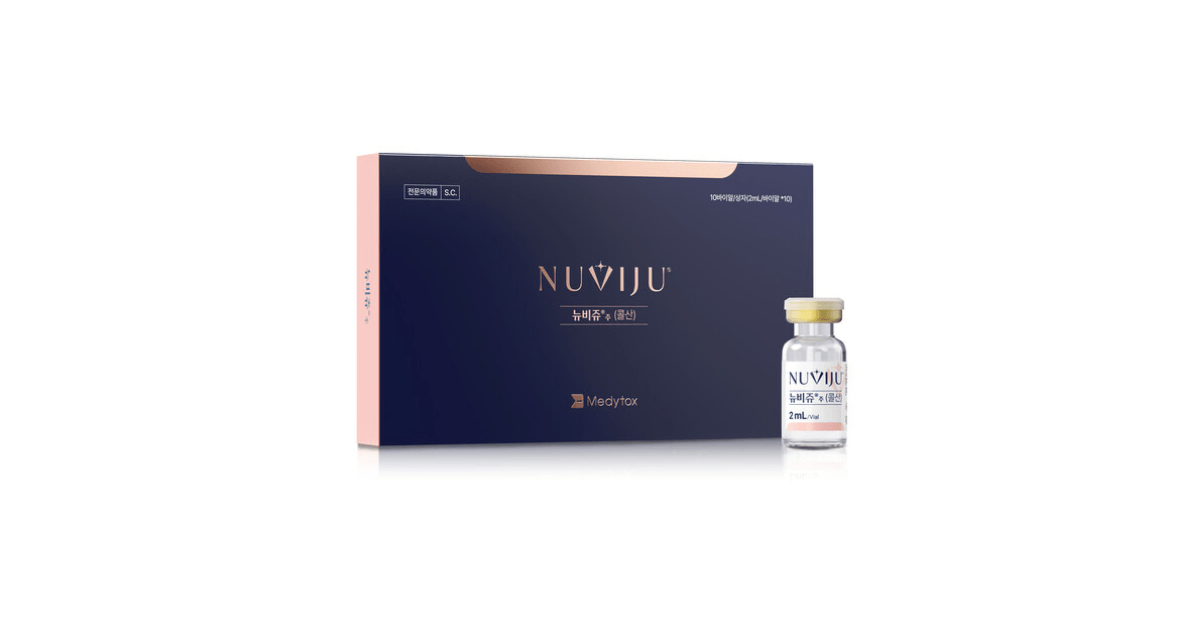
Medytox said it will launch Nuviju, a fat reduction injection for submental fat, on March 30, marking its entry into the medical aesthetics segment.
Nuviju is the first domestically developed fat reduction injection using cholic acid as its main ingredient, and has been designated the 40th homegrown new drug by the Ministry of Food and Drug Safety.
The product is indicated for the treatment of moderate-to-severe submental fat, commonly known as a double chin.
Unlike existing products based on deoxycholic acid, Nuviju uses cholic acid, which the company said may help reduce side effects such as swelling and bruising. It is also formulated to match physiological pH and excludes certain chemical excipients associated with irritation.
In a phase 3 clinical trial involving 240 Korean patients, 88.6 percent showed at least a one-grade improvement on a five-point scale compared with placebo, while 46.7 percent achieved at least a two-grade improvement.
Medytox said it plans to leverage its network of hospitals and clinics to support the product’s launch and expand its presence in the domestic aesthetics market.
The company also aims to create synergies with its existing portfolio, including Coretox, Newlux, and Neuramis.
A Medytox official said the company would focus on strengthening brand awareness and expanding adoption following the launch.