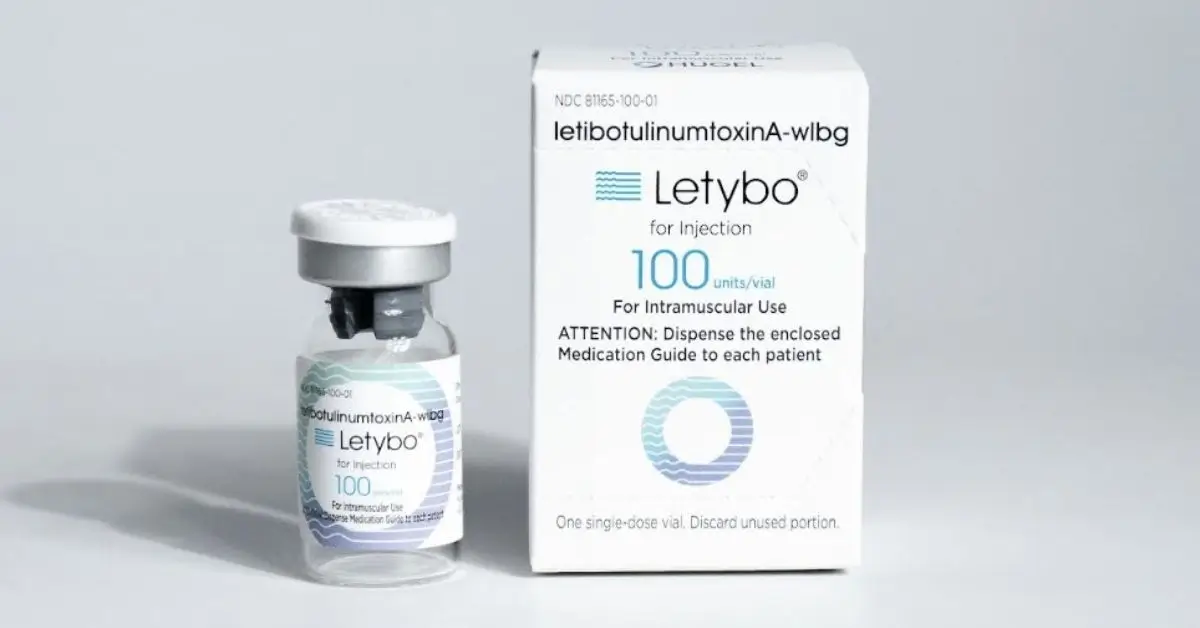
With proven results backed by extensive clinical research, Letybo is a globally trusted brand for over a decade, ranking as the #1 neuromodulator in South Korea. With over 29 peer reviewed studies on the active ingredient letibotulinumtoxinA, it’s approved in over 65 countries with over 31 million successful treatments to date.
Letybo is now fully commercialized in the U.S. through the partnership between BENEV and Hugel, marking a significant milestone in the neuromodulator market. As only the 6th neuromodulator to gain FDA approval in more than 20 years, Letybo offers U.S. providers a trusted solution for glabellar lines backed by robust clinical data and a proven global track record.
“We are honored to join forces with Hugel to bring Letybo to the U.S. market,” stated Ethan Min, Chief Executive Officer at BENEV. “This partnership with Hugel allows us to strengthen our market position and fulfill our mission to BENefit EVeryone through transformative aesthetic solutions.”
According to Dr. Hyoung Jin Moon, Representative Executive Officer of Hugel, Inc., “We are delighted to embark on the Letybo journey with BENEV. Our shared philosophies, including a commitment to provide premium products and education, science and technology, position us to offer a compelling service to the market. This partnership with BENEV will solidify Hugel’s position as a leader in the global medical aesthetics market.”
BENEV has been at the forefront of introducing science-backed aesthetic advancements from South Korea – a global leader in beauty innovation – to US aesthetic practices, and Letybo joins BENEV’s impressive portfolio of K-beauty inspired solutions.